
This is the homepage for eEF1A2 information hosted by Cathy Abbott’s lab at the IGC, University of Edinburgh. The gene encoding eEF1A2 has been found to be mutated in individuals with intellectual difficulties, often intractable epilepsy and sometimes autism. We want this to be a useful resource for scientists, clinicians and families. You can follow us on Twitter @CathyAbbottLab . Below are the answers to some frequently asked questions; more information is available on other pages.
What is EEF1A2?
At first sight the name is seemingly random letters and numbers, conveying almost nothing. The letters do in fact stand for something (eukaryotic elongation factor 1 alpha 2) but that’s not much help.
The EEF1A2 gene is the code for a protein called eEF1A2. This is, in turn, responsible for making all other proteins in nerve cells and muscle. The process of making new proteins is called translation, and all cells completely depend on this process. When eEF1A2 goes wrong, it will affect how well the nerve cell and muscles can work.
We now know of ~100 individuals worldwide who have a mutation (i.e. a change in the DNA) in EEF1A2. Most of these are children or young people, but there are some adults. The only reason for there being so many more children is because the huge leaps forward with genetic diagnosis over the last few years have mostly been implemented in paediatric settings. It doesn’t mean that having a mutation in EEF1A2 affects survival. There are undoubtedly many adults with EEF1A2 related disorders out there, they just haven’t had the appropriate genetic tests.
Each of the mutations in EEF1A2 that cause epilepsy and/or developmental delay change just one of the amino acids (“ building blocks”) out of nearly 500 amino acids that go to make up the eEF1A2 protein. Each of these building blocks seems to be really important for the normal function of eEF1A2 in nerve cells. More information can by found on the page eEF1A2: a Primer
Why is there so much variability in how people are affected by EEF1A2 mutations?
There is a big range in development and seizure incidence amongst people with mutations in EEF1A2, and at the moment it is very hard to predict how any one person will experience the disorder. Why is this? Well, the most obvious answer is that there are many different mutations in EEF1A2. Different mutations affect the resulting protein in subtly different ways, some worse than others. There is more on this below. As there are so many different mutations, over 40 so far, any given mutation (e.g. E124K) has often only been found in a few children, making it hard to generalise.
Another potential cause of variability is that almost all the mutations we know of in EEF1A2 are what is called “de novo”. This means that they weren’t inherited from either parent but are the result of a random event. This could have happened very early in development, so that every cell has the mutation. However, if the mutation happens after the embryo has started to form, an individual could have a patchwork of nerve cells, some with the mutation and some without. This would likely mean that they were less badly affected than someone with a mutation in every nerve cell.
How might different mutations in EEF1A2 result in different degrees of severity?
As we all know, we need eEF1A2 protein to keep us healthy, and we all have two copies of the gene that makes it. Let’s imagine that instead of genes, we each have to take two doses of eEF1A2 each day to stop us from getting epilepsy- picture two of those drug capsules with little beads inside (see the picture below). In this case, the little beads are all the molecules of eEF1A2.
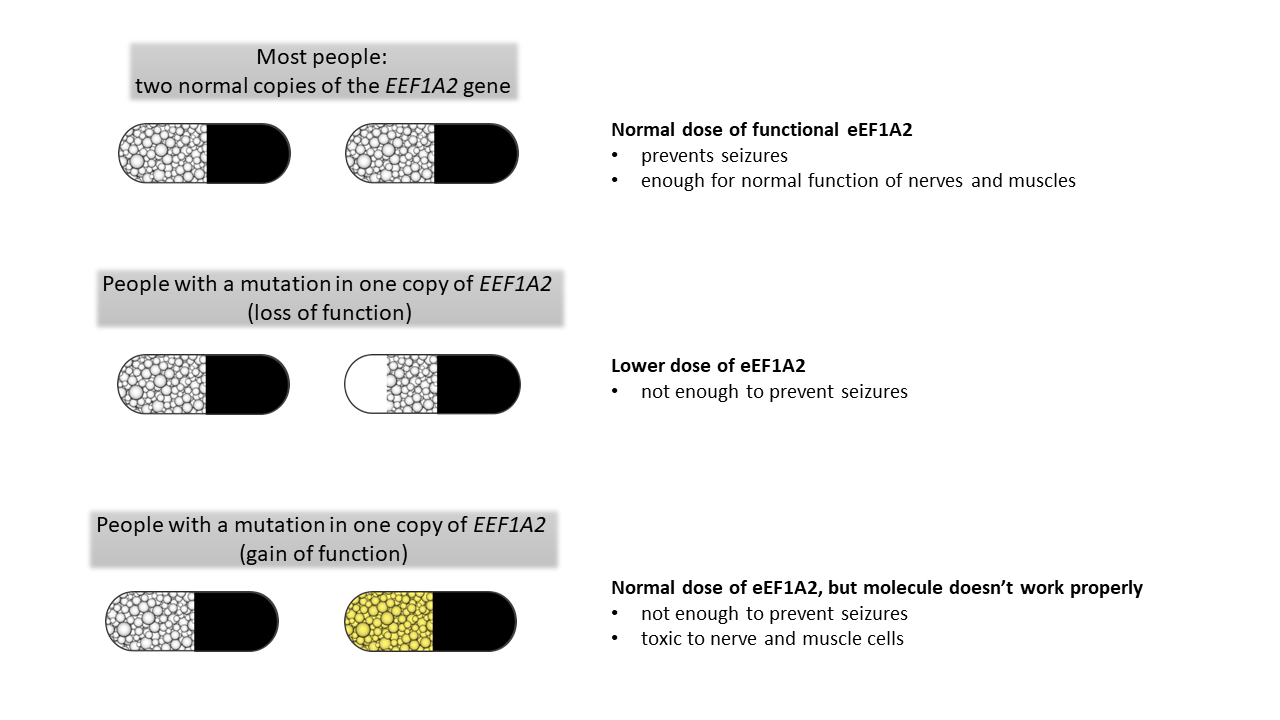
Now imagine what happens if you have a mutation in one copy of EEF1A2. You still have one capsule a day that works well (the “normal” copy of the gene that gives lots of normal eEF1A2 molecules), but the other capsule is not quite right. There are lots of different ways in which the other capsule can be not quite right (the different mutations), and the end effect will be different.
Some people might have a mutation which means that the second capsule is just not quite full enough (what we would call a “loss of function” mutation). This means that they don’t have quite enough eEF1A2 to prevent seizures, but do alright otherwise.
Other people have a mutation which means that the second capsule is full of eEF1A2, but it’s a version of eEF1A2 which is not quite the right drug. In this case, not only does the eEF1A2 not work well enough to stop seizures, but it is actually a bit toxic and can harm nerve and muscle cells. These people might have additional problems like intellectual disability and hypotonia.
There are lots of possible variations of this scenario, with different combinations of loss and gain of function. Some people will even have mutations in some of their nerve cells but not all, and this could affect whether they develop epilepsy, too. EEF1A2 disorder is a challenging to understand, but research is moving us forward all the time.
Where can I get support?
Any families who would like to be on contact with others who have children with mutations in eEF1A2 are welcome to join a Facebook group set up by Kari Haldeman. Kari has a child with the condition- you can find her at karihaldeman23 at verizon dot net.
How can I help with research?
Registries are really important for research, so that we can begin to look at the different mutations and collect information on their effects. In this way we will be better able to predict which mutations are likely to be milder and which more severe. With your help, we will be able to track outcomes, pull together information about which drugs help and which don’t, and ultimately design better therapeutic strategies and develop new drugs. Registries are also used as a way of recruiting patients for clinical trials. We are awaiting news from Invitae about the future of the EEF1A2 registry.
Why does Google give links to eEF1A2 and cancer?
This is a quick note for anyone new to eEF1A2, and especially anyone with a newly diagnosed child. If you do a google or other search for eEF1A2 you will find lots of papers talking about the role of eEF1A2 in cancer. This is not connected in any way with the mutations that cause epilepsy and ID and/or autism. The role of eEF1A2 in cancer relates to it being turned on inappropriately in cells other than neurons; this is essentially the opposite mechanism to that seen with the missense mutations.

So happy so see this site. My daughter was diagnosed with a mutation in EEF1A2 a year ago. We were told she was only the 5th in the world.
Hi everyone! Hi Kari! Does anyone know of a center that runs experimental clinical trials for this condition? Also, could you give me the name of a neurologist/epileptologist with expertise in this area?
Hello- I don’t know of any clinical trials for EEF1A2 specifically I’m afraid, though there may well be some for early onset epilepsies.
Thanks very much Kari- if any other parents come across this site please feel free to get in touch, we may be able to set up some sort of discussion board if that would be of any help.
hi, i am writing for a good friend of mine, she does not know English. her 1 yro daughter was just diagnosed with eef1a2 ;(
i told her I’ll contact y’all.
we are clueless as to this mutation. where do we go from here? what do u suggest to do next?
any info or direction would be a great blessings.
many thanks
T.
Hi Tirza,
I think the main thing for your friend to remember is that there’s a wide range of severity with eEF1A2 mutations, so it can be hard to predict the course for any one child. This is probably because different mutations in the same gene have different effects. There’s a Facebook group that Kari has set up- it’s in English but perhaps you’d be able to translate for your friend? That would be the best source of support from other parents, and there’s definitely some very positive stories. Good luck to your friend, and I’d be happy to try and answer any questions about the gene.
Cathy
Hi Tirza,
Welcome. Its hard when they are young. Where are you and your friend located? There is really nothing to do different than before she knew what the cause was. So much is still unknown. Therapies to help development and controlling seizures are the biggest things that will help. I am happy to add you both to the Facebook group if you are interested. Just leave me the information I would need to find you on there. What sorts of issues in the child dealing with? Hope to hear more from you soon.
Kari
My 3 year old also has EEF1A2 mutation. I’m very interested in finding other families through this!
Us too. Thanks for putting this site up.
You’re very welcome. I’m happy to answer any questions I might possibly be able to help with. I know that Samantha and Kari are keen to make contact with other parents, too.
Hi, thank you so much for coming back to me so quickly, I really appreciate it. I have many questions! 🙂 Please give me a couple of days to get to grips with the subject and I will come back to you with once I have put them into some order and I am not wasting your time with things I could have found out for myself and I am not peppering you with poorly thought through questions.
No problem, just take your time 🙂 I’m not a clinician, I should say, but am happy to help all I can. Incidentally, we know that there are at least four children in the UK with mutations in eEF1A2 identified through DDD, so you should be able to contact them too in due course.
Hi Samantha and Kari, we would be happy to meet up – we live in Chelmsford, Essex. Martha is seven and we just got the diagnosis from the DDD project [http://www.ddduk.org/] via GOSH genetics department. You can get in touch with me at alistair dot tweed at portigon dot com or you can send me a friend request on Facebook.
Hi Alistair,
I sent you a friend request. I started a fb page for eEF1A2 but it’s not active as no one has found it yet. My daughter is 9. We are in the United States. I think the testing program they’re doing over there is great!
We were just given this diagnosis today for my 22 month old. I know this is 5 years after the original post and comments, but I would like to be added to the Facebook group if there still is one. Thank you!
Comments are still active Kristen, and so is the Facebook group- nice to hear from you. Try searching for Kari Haldeman if EEF1A2 doesn’t work, and if all else fails just get back to me!
Hi, as promised, I have a list of questions and would be really grateful for any assistance you offer in helping me understand more.
Exome Sequencing results:
Roughly how many cases of EEF1A2 have been identified in total?
What is the age range of the people effected?
Are there any of them males?
Wasted Mice:
Please could you explain the differences between humans and wasted mice?
Is it that wasted mice have a complete lack of EEF1A2 and human cases do not?
Have there been any intervention studies in wasted mice to add the missing correct form of the protein?
Are there any planned?
Working backwards towards intervening and trying to alleviate the effects by adding the missing protein:
Does the protein cross the blood brain barrier?
Does the protein make it past the liver?
Does the protein get digested in the gut or does it get absorbed without being broken down?
Does it exist in any food sources in significant amounts?
If not, can it be manufactured?
The future:
Are there any promising innovations, technologies or strategies that I should be aware of or following?
How far off are gene editing and gene engineering technologies like CRISPR from being able to alter genomes to fix the EEF1A2 mutation?
Many thanks in advance for your time.
Warm Regards,
Alistair
Hi Alistair,
Right, here are some initial responses. Some may take a bit more thought, these are all excellent questions!
Exome Sequencing results:
Roughly how many cases of EEF1A2 have been identified in total?
We know of 12 so far, but this is likely to increase a lot as exome sequencing becomes the norm, and as eEF1A2 becomes included in specific screening panels.
What is the age range of the people effected?
In the literature the youngest reported was one, and the oldest is now in her early/mid twenties. This doesn’t mean anything in relation to eEF1A2 though, it’s a reflection of the screening programs and their emphasis on children whose families want a diagnosis. It does mean that we don’t have any information about longer term effects of the mutations, though.
Are there any of them males?
Yes, at least two are male- it looks rather skewed at the moment but it’s too early to deduce anything concrete, and there’s no expectation of a gender bias from the basic biology.
Wasted Mice:
Please could you explain the differences between humans and wasted mice?
Is it that wasted mice have a complete lack of EEF1A2 and human cases do not?
Yes, that’s exactly right. The mice that carry the wasted mutation (so one mutant copy and one normal) survive completely normally and have the same strength and coordination as non-mutant mice. They have no sign of seizures, either. However, we don’t know if this is because there’s a difference between mice and humans in terms of whether they can tolerate having a 50% reduction in eEF1A2, or whether the reason all your children are affected is because the mutant protein which is being made is toxic in some way. This is a crucial question that we have to answer before we can do anything else, as it goes to the heart of any treatment. If the mutant protein is toxic, then the last thing we should do is to treat with anything that increases the level of expression. On the other hand, if the epilepsy and so on results simply from a lack of eEF1A2, then replacing it would be the way forward.
Have there been any intervention studies in wasted mice to add the missing correct form of the protein?
Are there any planned?
No, not the protein because it’s incredibly difficult to deliver a protein to the brain, I don’t know of anyone who has even tried this. We have corrected it genetically, but that’s not an option in humans.
Working backwards towards intervening and trying to alleviate the effects by adding the missing protein:
Does the protein cross the blood brain barrier?
I don’t know of any proteins that have been shown to cross the blood brain barrier, only small peptides, but will think about this.
Does the protein make it past the liver?
Does the protein get digested in the gut or does it get absorbed without being broken down?
Never been tested but unlikely to survive I’d have thought.
Does it exist in any food sources in significant amounts?
No, not at all I’m afraid.
If not, can it be manufactured?
Hmm, I guess so, although it would be very hard to make a properly folded and processed protein with current technology. Would be worth considering if there was any prospect of getting it into the brain, but I think approaches using small molecule drugs to try and upregulate normal eEF1A2 or downregulate mutant eEF1A2 (depending on the mechanism) will be a more productive strategy.
The future:
Are there any promising innovations, technologies or strategies that I should be aware of or following?
Well, small molecule screens as mentioned above, and also CRISPR. I would keep an eye on what’s happening with Rett syndrome, as that’s a good example of a dedicated and mobilised body of parents and scientists working towards therapy.
How far off are gene editing and gene engineering technologies like CRISPR from being able to alter genomes to fix the EEF1A2 mutation?
A long way off as things stand, simply because of the problems of accessing the brain, but things are moving incredibly quickly in this field.
I hope some of that is useful, but feel free to keep asking meanwhile!
Cathy
Thank you so much for your time and help, Professor Abbott. Very much appreciated. 🙂
You’re welcome. I should have said, when I talked about wasted mice, I’m making a distinction between the mice that carry the mutation (heterozygotes, more like the situation in humans, with 50% of the normal levels, and normal survival) and wasted mice themselves, which have no eEF1A2 at all and don’t survive. That’s probably just made it worse…
Hi guys. My 6month old son was recently diagnosed and I am interested in connecting with other parents. We are in the U.S. But emails can travel anywhere. My email is MrsHarper122 at outlook dot com Thanks so much and I look forward to connecting with u guys!
Hi! Glad to hear from you. I’m going to email you.
Hello all – having taken part in the DDD research study – at last today we have received the EEfiA2 diagnosis four our son Arlo aged 11. We are keen to connect with other families like us – we live in Brighton UK and have lots of questions plus hopefully some info to share about our son’s experience so far. We would love to hear from you!
Hello Keely,
It’s good to hear from you. I’m sure other parents will get in touch very soon, and of course I’d be happy to answer any questions if I can be of any help.
With best wishes,
Cathy
Thank you Cathy – so lovely to get a response so quickly – still absorbing the information but excited to start the journey and find out more. I am sure I will have many questions in the weeks to come. Best wishes Keely
You’re very welcome, any time! 🙂
Hi Cathy – our Son has atonic siezures – and what we call head nods and eye rolls – this is now well managed with medication but I wonder if all others diagnosed with eef1a2 all have the same type of siezure?
Hi Keely,
I’m not a neurologist, but we are writing a paper about the latest case studies at the moment, and essentially the type and severity of the epilepsy is very variable. There are some children who are like Arlo, in that their seizures are well controlled, but others who have tried multiple types of anti-epileptic drugs without success, and who have tonic and tonic-clonic seizures.
There aren’t enough cases yet to know whether this comes down to exactly what mutation different children have, but that certainly seems to be part of the story. When we look at the eEF1A2 molecule, the mutations that give milder epilepsy and hypotonia seem to be further away from all the crucial parts of the protein. Hearing from families is such an important part of the puzzle for us, so thank you all for being so generous with your time!
Cathy
Thanks for your reply – happy to share any information that might help other families. We are so lucky to have found a medication that works for us after much trial and error maybe others with similar seizures will find it works for them too. Thank you for your help – I am so happy to find this website I thought it would be a struggle to find any information – this is an exciting day for us it is great to have the chance to make contact with other families facing similar struggles.
Thanks again – Keely
I’m so pleased it’s been useful!
Hi Cathy, when we received our results I included a link to this website to my FB page to help to explain to friends and family about Arlo’s diagnosis as we found your information so helpful and I am not great as explaining as still absorbing the information myself. It has also been an amazing help to us to find a way to contact other families in our situation. I hope that is OK as I appreciate it was not intended as a social forum – if you would like me to remove the link please let me know. Thank you again for publishing this information about eEF1A2. Best wishes Keely
Hi Keely,
Oh, not at all, I’m delighted it’s being used in this way. I deliberately set it up so comments could be made. I have it so that I moderate commments just in case it gets spammed, but I’m more than happy for anyone to post here!
Hope you’re having a good weekend,
Cathy
Thank you Cathy that is very generous – thank you for your support
kelly i want to contact you. And other familly. +905525225723 my phone number can you write me on whatsapp
Hi Keely. I’m Kari. My daughter Kristin is 10. She was diagnosed a year and a half ago. It’s great to meet you. Glad you finally got a diagnosis! Love to hear more… I also started a fb page if you’re on facebook.
Hi Kari, it is so great to hear from you! I would love to join your FB page – I searched but only came up with my own post – if you search for Keely Lewis Brighton hopefully you will find me.
Arlo is 11 and a half, funny, strong willed, happy and beautiful!! He was diagnosed with epilepsy as a baby and has severe learning difficulties. He is non verbal and didn’t walk until he was almost 6 years old – (now climbing everywhere!!). I would love to learn about Kristin – I will look out for your FB page.
Keely,
I’m pretty sure I found you but it won’t let me add you for some reason. I’ll try on my computer when I’m home later.
Great I will look out for you!
Think I found you on FB – have sent a message
Keely and Ben such amazing parents wishing you and your beautiful boy all the best. Paul, Carlos and Jacob.
Hello to everyone. Talking from the UK here. My 3 week old daughter has been suffering from seizures since she was born on 02/09/2015 and me and my wife have been told today she has a deletion of 20q 13.33 chromosome. Mutations of EEF1A2 & RTEL1. She also has deletions of CHRNA4 & KCNQ2. She was born with absolutely no de-formalities. Time will tell for her development. I am yet to find any information about a patient with 4 mutation/deletions.
Hello Samer,
Thanks for your post- this must all be very new and scary. You’ll get some fantastic support here, I am sure.
Best wishes to you all,
Cathy
Hello Samer,
We have an 11 year old son with EEF1A2 epilepsy, we have only very recently received a diagnosis. I understand how important it is to feel you can contact other families who’s children share the same diagnosis and how lonely it can feel when you don’t know anyone who shares your experiences. It must be a very stressful time for your family, coming to terms with with having a child with health concerns and witnessing them suffer seizures is a lot to cope with I know. I am so sorry not to be able to offer any advice or experience relating to your daughters particular diagnosis.
When we received our sons diagnosis we were given the following details of organisations who may be able to help and offer support in finding other families experiencing similar struggles. I am sorry if you are already aware of these but if not I hope they may be of some help to you.
Best wishes to you and your family
Contact a family have a lot of helpful information on their website.
http://www.cafamily.org.uk
Also SWAN UK
http://undiagnosed.org.uk/
which supports families without a diagnosis.
There has been a lot of research recently and currently, families who have had many years with no diagnosis are receiving results and are newly able to give a name to their childs condition. It is possible that SWAN may be aware of specific family support groups starting up for previously undiagnosed conditions.
Hi Samer,
I was sure I replied to this but something must have gone wrong. It sounds like your child probably has a chromosome mutation while most of us it’s just a single gene. I think it’s great they can tell you which specific genes are affected. I started a fb group if you’re interested.
Kari
You’re absolutely right, Kari.
It’s difficult to disentangle the effects of deleting eEF1A2 in cases like your daughter’s, Samer, because there are two other epilepsy causing genes so close by (CHRNA4 and KCNQ2, which is right beside eEF1A2). There are certainly other cases in the scientific literature, but of course the exact size and position of the deletion can vary from child to child. I hope things have settled down a bit for you, and that you are getting support- Kari’s FB group is great.
Hi Kari. Would def be interested in the FB group. Add me if you can. My name is Sam Oujeil in Bristol, England
My daughter was diagnosed a month ago with a eef1a2 mutation. She is 3.5 with seizure disorder, global developmentally delay, Microcephaly, cvi, delayed myelination, thinning of the corpus callosum, low muscle tone, and feed difficulties.
Hello Mandi,
Thanks for your post – I think you’ve already found the Facebook group where I know you’ll get lots of support but feel free to ask any questions here too and I’ll do my best to answer them!
Cathy
Hi, our son Noah has tested positive for mutation of the EEF1A2 gene.
Very similar symptoms as your daughter.
Hello Stuart, glad you found the website. Kari has set up a Facebook group as you may have seen, with lots of shared information.
Best wishes to you and Noah,
Cathy
Just an update regarding my daughter. So after 9 weeks from leaving hospital without any seizures, they began once again with 2 a day for around 2 weeks, we upped her meads of Topirimate and she stopped. Since then we have discovered she also cannot break down milk proteins and is now on Nutrimigen, she is a different baby. She has lots of gene deletions including EEF1A2 and RTEL1 but is flourishing. She is currently showing no signs as of yet in developing her instincts. She is rolling over, playing with toys, eating well, not talking much but sometimes, focuses well, reacts to sounds and most of all screams like a real baby. There is hope, and I wish everybody well for the new year.
Sam
That’s great news Sam, I’m so pleased. The situation with the children with missense mutations and no deletions is probably different, but I know some children with the missense mutations also have food intolerances. Hope things continue to improve for your baby!
What is the name of the FB group?
Thanks,
Stuart
Hi Stuart,
It’s just called EEF1A2. Kari has posted a welcome to you there, so you should be able to see it in your notifications.
I added you so you should be able to access it. Let me know if you can’t. I tagged you though so it should show up.
Kari
Cathy, Do you have any recent information about the number of kids diagnosed with a mutation in eEF1A2 now? Anything new going on with the research side of things that you want to share? Thanks. I am so grateful that you set up this website. It seems to be a good way to find new families.
Hi Kari,
You beat me to it, I was just thinking this morning that it would be good to collate all this information, and ideally have a table on this website about the different mutations. It’s still too early to know whether specific mutations will predict anything about the clinical picture in individual children but it would be well worth starting to look.
There have been three more cases published in the last month, and 6 more on a poster at a big US epilepsy conference, plus the 6 (including some of you!) in the paper we have submitted. There are almost certainly people in the Facebook group who are in addition to this (it can be hard to tell as the publications are obviously anonymous, but two of the three recent published cases are in Japan). So at least 20 with the 4 already published.
I’ll aim to post a proper update (not just in the comments) in the next week or so. If people would be willing to share the details of their children’s mutations then we could start to build a database that new parents could check. There are now at least 4 individuals each with G70S and the same with E122K, certainly, so patterns might emerge.
I’ll update more with our work too!
Cathy
That sounds great! I’ll ask everyone to post their info on the fb group. Look forward to your update and to reading the paper when it’s published. Thanks!
Kari
Fantastic, thanks Kari. If anyone wants to share details with other parents but would prefer that I didn’t use the information in any database then they can just say so, I’ll obviously honour those wishes!
Hi my son was just diagnosed with eef1A2 with the help with his doctors at Kennedy krieger institute he 16 years old I thank God for his doctors for founding what is wrong with him with live in the united states he is the first case for the doctors here
Welcome Kathy, I’m glad you found the site. If you are on Facebook and would like to join the group there I know you’d be very welcome. Your son is a but older than many on there so I’m sure the other parents would be keen to hear how you are all getting on.
HI Kathy. If you are on Facebook please join our page. EEF1A2. My daughter is 11 and we live in Delaware. Glad you found us.
My son was diagnosed today with EEF1A2… really wanting to learn more
Hello Robin! Happy to answer any questions, and glad to see you’ve found the Facebook group.
Yes it has been very difficult for my son and i .I think he has had it for years and we just found out ourself he is so much better now that the doctors have hom on the right medication.
robin can i contact with you please write me on whatsaap +905525225723
Can you have this gene issue and only have intractable epiiepsy without the intellectual disabilites and autism.My son just did the mouth swab no results yet.Hes verbal not dejayed just started seizures in Aug.I have surgery controlled TLE 29 yrx seizure free.I was intractable 3 yrs before surgery at 15.I havent been tested yet
Hello Renee, I’m not a clinician so don’t take this as professional advice, but in all honesty we’re just scratching the surface of the effects that mutations in this gene can have. There are definitely people with autism and ID but not epilepsy so no reason to suppose that it’s impossible to have epilepsy without the other aspects. This is definitely true of other genes, too. Good luck with your results!
Hello,
Our 3 year old son has been diagnosed with a de novo heterozygous variant in the EEF1A2 gene, c.43C>G p.(His15Asp) after performing a whole exome sequencing. He has epilepsy, hypotony, developmental delay (he still does not walk or talk). He is on several medication to treat the seizures and we work tirelessly with the therapists to improve his motor and intellectual skills.
Our son was born in the US (in Nevada, as we lived there for 4 years) but we are from Barcelona (Spain). We’re really glad to finally have a diagnosis but we’re desperate for more information, to get in touch with people in the same situation, news on treatments, etc.
Thanks for putting this site up!
Hello, thanks for getting in touch! Your son has yet another new mutation that we have not seen before. There is no news on treatments at the moment but there is a fantastic EEF1A2 support group on Facebook with lots of families in similar situations. I think Kari Haldeman will see your comment here and can add you to the group- I am also a member so if you want to ask any questions about research please feel free.
Cathy
hello I’m a speech and language pathologist. My client who 22 montl old baby boy has this gene mutation which uniqe one. I will researvh it academically and effects on communication. I could not found facebook page. Please share it to me and adding me in this group I will be pleasure being in contact and share knowledge each other. I’m live in Türkiye.
Y.
Hello- thanks for getting in touch. Kari administers the Facebook group and should be able to add you.
Hi. Please feel free go post or ask questions on the Facebook page. There is still very little known about the gene and mutations outside of seizures and low muscle tone.
Kari
Hello
We are the parents of a thirty year old daughter who was diagnosed with EEF1A2 several years ago. Today we had some updates from our clinical geneticist and searching on the internet I found this forum. When our daughter was diagnosed in 2012 it was hard to find any information or parents who had a child with the same disorder.
We live in the Netherlands and as far as I know in our country there are 3 diagnosed with this gene.
I will be glad to give more information if anyone is looking for it. I also hope to find some more background information myself.
Thanks
Annie
Hello, good to hear from you. I think your daughter was the start of this whole story! If you would like to join the Facebook group you would be very much welcomed, and meanwhile I am very happy to answer any questions. Cathy
Thank you Cathy
I have joined the face book group and introduced myself and my daughter. If I understand well that within the gene EEF1A2, there are mutations, so different disorders. Am I right?
Are there known regressions in developments? Or in motoric? With My daughter it seems like her walking is getting slightly worse. It is always hard to tell what is due to epilepsy or medication.Her medication is changed in the past year.
Annie
That’s great Annie- your daughter is beautiful! We still don’t know exactly how much difference the different mutations make, as there are so many that have only occurred once or twice. I would say probably all the same disorder but different severities, though that is only an informed guess. There is no evidence for regression but your daughter is one of the oldest people known with epilepsy caused by a mutation in EEF1A2. As you say, hard to know what is down to medication.
My granddaughter, now almost 1 year old, was recently diagnosed with EEF1A2. Apparently she has a brand new mutation. I have been searching for information in order to help my family learn more about this. Thank you so much for creating this place for information and support. I will be researching some of the websites you have posted. We live in the U.S.
Hello Terri – you’re very welcome. I’m more than happy to try and answer any questions about the research and you and your family might like to join the Facebook group for support from other parents and grandparents? With best wishes, Cathy
Wow, this is a great resource, thank you so much for setting this up! I have my homework cut out for me as I just found out days ago that my 5yo daughter has a EEF1A2 mutation (variant p.D252H, coding c.754 G>C), not even sure what that means yet. If there are any other resources please post! Thanks again! PS- I already emailed you directly after seeing the FB group =)
Thanks, so good to hear from you and very glad it’s useful!
My daughter, now 27, has recently been diagnosed with an eef1a2 gm. She is the most wonderful human being I have ever known! How many cases are now identified?
How lovely to hear that! I can’t tell you how many in total because the numbers seem to be shooting up, but I would say at least 70. There will be loads more who just haven’t had genetic testing, though, or whose information hasn’t been described online anywhere. Your daughter is one of the older people we know of with a diagnosis. Do join the Facebook group if you haven’t already, there are lots of lovely supportive families there, but feel free to ask any questions about the science here, too! Cathy
Hello everyone!
My niece was diagnosed with this eef1a2, she is 3years old and she lives is Romania, she had the diagnosis from Italy and she got some medication from there Subril and Desparchine.
The chances for a corect medication are not high, I was wondering would UK be a chance for her? If there’s any medication or special treatment or something to help her out.
Many thanks
Hello, and thanks for your comment. I am not a clinician so can’t help with medication advice I’m afraid (although experiences seem to vary a lot between children and may depend on the exact mutation). There’s a Facebook group where other families may be able to share experiences that would help, by all means email me or Kari if you’d like to be added. Good luck, Cathy
Thank you for explaining this disease a little better and for all your helpful sites. It is so hard to see our Jax Cunningham struggle so hard to fit in. He had such a sweet spirit and sometimes he seems so alone I pray your research goes well, and you will make quick progress to help all these precious children. Pat Branch-Fontaine. Jax is my grandson.
I am so sorry this comment got lost. It’s lovely to hear from you and thanks so much for your kind comments.
Hello
My daughter has recently diagnosed with eef1a2 and i heard about this fb group,is this still activ? Cause i can’t found it ,i added someone called kari but i don’t know if this the same one u r talking about or not
Hello Sara, yes, the FB group is active. If you have any problems I am sure Kari would respond by email- good luck.
Hi Sara,
It’s Kari. Try searching EEF1A2 all caps on fb. Or look for Kari Haldeman. I live in Delaware in USA. My profile picture is my daughter with a rare disease banner around it.
Do u have any information about this variant NM_001958.2:C.271G>A EEF1A2
Because i didn’t find anything with the same sequence and the report does not provide me with anything
The report was with same gene but different variants and they can not detect the specific function of this one listed
Hello Sara. Yes, there are two other D91N (this is the change at the protein level that results from C271G>A) cases described, one in our paper Lam et al https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4947865/ and one in the one by Carvill and colleagues that Danielle Boyce Reinhart posted on here, both teenagers now.
Hi Team,
Recently we did genetic test for my daughter and found below details as a result.
We really need your support and suggestions. Could you please help.
Gene (Transcript) # Location Variant Zygosity Disease (OMIM) Inheritance Classification
EEF1A2 (-)
(ENST00000217182.6)
Exon 4 c.374C>A
(p.Ala125Glu)
Heterozygous Developmental and
epileptic
encephalopathy-33
Autosomal
dominant
Uncertain
Significance
Hello Prasad,
Thank you for sharing your daughter’s results. I am not a clinician so can’t give medical advice but I can tell you that your daughter’s exact variant, A125E, has been seen in a couple of other children so it is almost certainly causative and not “uncertain significance”. If you are on Facebook I would really recommend joining the EEF1A2 group as there are lots of other parents there sharing their experiences with their children, including which meds they have tried. I am happy to try and answer any specific science questions you may have, too.
Best wishes,
Cathy
Thank you Cathy. Is there parament solution or cure for this problem?
I’m afraid not, so far, though we are working hard on understanding it more with the aim of improving therapy, at the very least. Different children seem to respond differently to the many anti-seizure medications so it is definitely worth hearing from other parents.
Understood, thank you Cathy.FYI(I already request sent to join the EEF1A2 group)
Hi Cathy,
(1) Is here a way to tell if your child has a low dose or toxic dose of EEF1A2, if so, are there benefits to knowing that info?
Also, our daughter’s mutation is EEF1A2 (c.754G>C), variant D252H. She has no history of seizure activity. (2) Are there other cases with no seizures or should we be more diligent in detection?
Thank you so very much in advance.
~ Virginia
Hi Virginia,
No easy way I’m afraid- we do lots of different types of experiments in the lab but focus on a few mutations for most of them. I suspect they will almost all be a combination of low/toxic and it’s a balancing act. The only obvious benefit would be once there are treatments, as they will probably target loss of function more easily, but could still be of benefit to all affected children. We will test any treatments in our mouse models so that will help to judge how wide the benefits will be.
D252H is one of the variants we know most about in the lab! There are two other individuals I know of with the same variant. One is still about 5 and hasn’t had a seizure. The other was described in a paper by Nakajima et al and she didn’t have a seizure until she was 8. The antiseizure medication worked well though so her seizures were controlled.
Hope that helps but feel free to ask any more!
Best wishes,
Cathy
Hello to the moderators,
My wife is Danish / me fr Cameroon and We live in Danmark . Our son has been diagnose with the EEF1A2 (epileptic encephalopathy) gene. He is just 4 months old. We are hoping to Connect with families having to deal with this and their experinces as the child grows up.
Merci
Fr Tambe and Mette
Hello to you both. The Facebook group is the best source of support and connection with other families. If you contact Kari Haldeman she will be able to add you. Meanwhile if you have any questions about research or science I will do my very best to answer. With best wishes to you all, Cathy
Buenos días,
¿El grupo de facebook sigue activo?
He intentado contactar con Kari pero no he tenido respuesta.
Soy española y me gustaría contactar con otras familias en esta situación.
Gracias.
Yes, it is active, I will mention your request to Kari.
Kari suggests sending the request again through Facebook, and remember to answer the question to get the request approved. Good luck!
Muchas gracias.
Hi! Is there a newsletter or group outside of the Facebook group? We live outside of Chicago, IL. Our 13 year old son was just diagnosed with this mutation. He is our 3rd of 4 kids. He has always been developmental delayed, with neurocognitive deficits, and has diagnoses of autism and epilepsy. His seizures were not diagnosed until he we 8, and they have gotten progressively worse over the years. He has partial complex seizures. He currently takes 500mg/day of Lamotrigine ER. He lives at a therapeutic residential school because he has a very difficult time with social emotional regulation, and is also very impulsive. Thanks so much, Abbie
Tengo un hijo de 9 años con una mutación patogénica en este gen. Me gustaría entrar en contacto con este grupo de pacientes.
Muchas gracias
I am so sorry, I completely missed your comment (I normally get email notifications). The patient group is on Facebook but do feel free to email if you can’t find it.
Would you please give again here a link on your group in Facebook. My son was diagnosed with EEF1A2, too. I am from Belarus.
I’m really sorry but I can’t give a link as it’s a private group. If you search on Facebook you should find it, and Kari or one of the other moderators should be able to approve your request.
Hi, my daughter, Isabella was recently diagnosed with a mutation in EEF1A2. Isabella will be six years old in July. As of yet, & quite strangely given she has a EEF1A2 defect, she hasn’t yet shown any signs of epilepsy or had any kind of seizure. I would be keen to discuss with others, particularly anybody who has a child that hasn’t shown signs of epilepsy or anybody’s child that has developed epilepsy in later years. We were told that most children with this defect start with epilepsy from around 4 months old. We live in Bolton (Uk)
Many Thanks
Adam
Hi Adam, good to hear from you. There are definitely children who don’t develop epilepsy, or who have seizures only later on. It may depend on the exact variant. It would be worth joining the Facebook group, this exact question has been asked before and got a lot of replies.
Best wishes, Cathy
Thank you for your quick response Cathy. I will definitely join the Facebook group.
Kind Regards
Adam
Sorry Cathy. Please could you send me the link to the Facebook page?
Thank you
Hi Adam,
Our son Sam is 14, and was diagnosed with a mutation in eEF1A2 when he was 12. He wasn’t diagnosed with epilepsy until he was 9, and I believe he had been having complex partial seizures for about 6 months before diagnosis. When we look back we think he might have been having short absence seizures for several years before this that went unnoticed. Sam has been developmentally delayed since he was a baby, so had always been under the care of several doctors and therapists and no one ever observed any seizure activity. I know the prognosis is different for every child, so hopefully your daughter won’t ever have epilepsy.
Hi Abbie. Many thanks for your comment. It is very helpful to know that there is still a possibility that Isabella may develop epilepsy. The picture that has been painted to us is that she is unlikely to develop it if she hasn’t by now. This clearly isn’t the case given your situation so thanks for clarifying this.
Would you mind telling me if your son has continued to develop & make progress. Isabella has had noticeable severe global development delay since being just a few months old. It took her a long time to be able to lift her head from a lying position & even longer to crawl. She is still unable to walk unsupported but is showing signs of progress by taking a few steps unsupported. She is also non-verbal. Are you aware of or has your son developed speech?
Kind Regards
Adam
Can I also ask, does anybody know the link to Kari’s Facebook page so I can join?
Many Thanks
Adam
It’s a private group Adam, so you just have to search EEF1A2 on Facebook and send a request to join, sorry.
👍🏻Thank you
Happy to answer questions. I am only on Facebook to be in the private group, but if you look up my name I think we can have a conversation through that.
Hi, my name is Françoise, my son Felipi is 14 years old and we are brazilian. We have investigated his development delay since jé born and only after doing an Exome and an Exome reanalys we found EEF1A2 disorder (among others). His clinical syntomes includes epilepsy (mioclonic since he was a baby and now after 14 years, presenting focal disperceptives seizures), severe global development delay, autism, absent speech and also he achieved walking at 7 years.
My main question at this moment is related to the medication used to treat this kind of epilepsy seizures. Also would like to know if is there some brazilian people with Eef1a2 disorder
Hello Francoise, good to hear from you. I’m afraid I can’t answer questions about anti-seizure medication but if you are able to join the Facebook group there are lots of parents there who will be able to share their experiences. I can’t think of anyone else from Brazil at the moment but again the Facebook group would be the place to look- good luck!
Hi, our son is now 12 years old. We got the diagnose 5 years ago and were told he was number 13 in the world.
And that there was only 1 child with the exact Same mutation as him .
This was a girl in the UK. She was I think around 11 years old at the time.
So now she wil be around 16 .
I would love to be in contact with her parents !
But doctors never gave me a name …
Hello! I don’t think doctors are usually allowed to give out names of other patients I’m afraid, but the parents may well be on the Facebook group so worth joining and asking there. If you don’t mind sharing which mutation your son has I am happy to tell you if I know of others with the same variant (you can email me if you’d rather not put it on here).
Best wishes,
Cathy
bonsoir,
je suis la maman de Louis 19 ans polyhandicapé diagnostiqué en 2012 du gène EEF1A2.
j’habite dans le Nord Pas De Calais.
serez t’il possible d’avoir des contacts avec des parents.
Hello Agnes. The best way to contact other parents would be on the Facebook group. If you have problems, search for Kari Haldeman. Bonne chance!
Dear Group,
first of all, i am grateful that there is such place we can share and support each other.
Yesterday, we were shocked by the news that our daughter (13 months) has EEF1A2 p.Gly70Ser. Naturally i started to research for info and was totally shocked and stressed for the potential development (or its delay).
I also tried to enter the FB group and sent a request – please approve it when you can.
And my biggest question is – i saw that the base of reported cases with this genome changed are extremely small (around 4 in most recent studies) and all of them are severe in terms of global development.So…is there some positive option for development (based on lack statistical cases and individual development)of what you know and have seen.
I believe(hope) that the individual development of my daughter should lead not to severe but more mild case – she has never had seizures, she develops her movements (we do Vojta, Bobat, HBO), she is social (smiles, hugs us while lying, laughing at her brother when he plays). .Yet, she has some delays – not crawling yet (but close), no words……but she sings:) and with different intonations.
I would be happy if you can share if someone had similar experience and what would be your advice.
Big thanks in advance and best regards,Hristo
Hello Hristo. I am so sorry you have been shocked. There are far more than 4 reports of children with the same variant as your daughter’s but in most cases seizures have started very early. It sounds as though your daughter is doing well and you are doing all the right things. The Facebook group will be a good source of support, I am sure.
Thank you for your prompt reply. I already sent a request for the FB – expecting to be approved:)
Final question – based on your experience and knowledge, do you believe it is possible to reach better outcome with this diagnosis (regular or close to regular – speech, walking independently and most importantly mild mental delay)?
Thanks again and see you in FB. have a good one. Hristo
I’m not responsible for the Facebook group I’m afraid but I am sure you will be approved soon. I have to be honest, I am not aware of anyone with this diagnosis having an outcome quite like the one you describe but I am not a clinician. Walking is certainly achievable for many though and the fact that your daughter is still developing is really good.